 
They can understand and predict how an element might react or behave in a certain situation.Įach element has its own name and abbreviation in the periodic table. This lining-up and grouping of similar elements helps chemists when working with elements. You can see all the groups in the table below. They are all very similar in that they have only 1 electron in their outer shell and are very reactive. Another example is the alkali metals which all align on the left-most column. They all have a full outer shell of electrons, making them very stable (they tend not to react with other elements). These elements all line up in the eighteenth or last column of the periodic table. 
One example of a group is the noble or inert gases. There are 18 columns or groups and different groups have different properties. 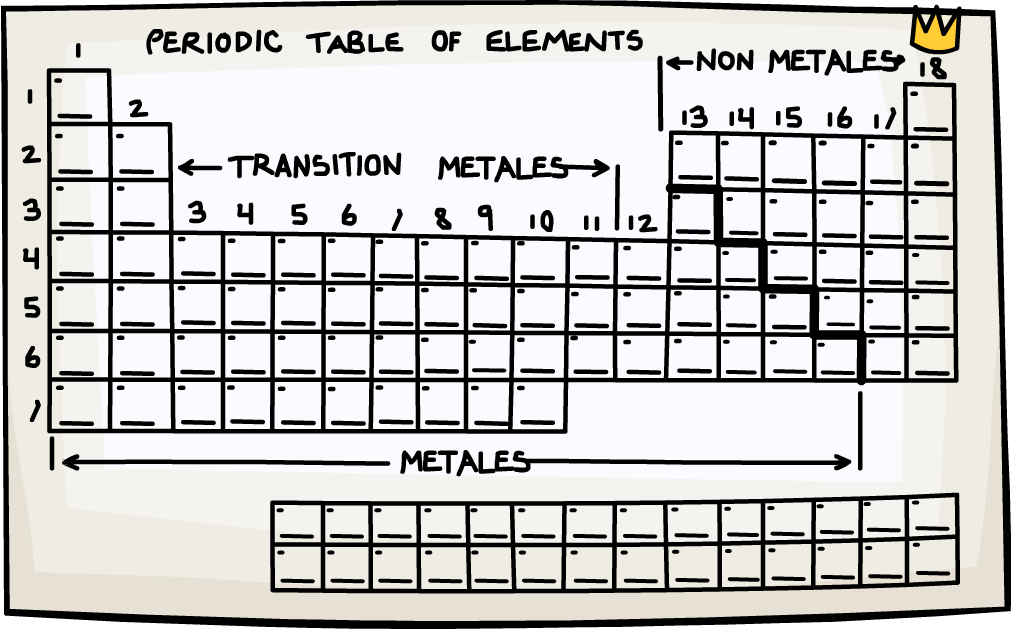
Groups are the columns of the periodic table. In each period the left most element has 1 electron in its outer shell and the right most element has a full shell. The first one is short and only has two elements, hydrogen and helium. There are seven (or eight) total periods. When they are lined up this way, elements in the columns have similar properties.Įach horizontal row in the table is a period. Some columns are skipped in order for elements with the same number of valence electrons to line up on the same columns. From left to right elements are lined up in rows based on their atomic number (the number of protons in their nucleus). Because its 2n shell is filled, it is energetically stable as a single atom and will rarely form chemical bonds with other atoms.It is called "periodic" because elements are lined up in cycles or periods. For instance, lithium ( Li \text Ne start text, N, e, end text ), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1 s 1, s orbital and eight fill the second shell-two each in the 2 s 2s 2 s 2, s and three p p p p orbitals, 1 s 2 1s^ 2 1 s 2 1, s, squared 2 s 2 2s^ 2 2 s 2 2, s, squared 2 p 6 2p^6 2 p 6 2, p, start superscript, 6, end superscript. Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
